|
7/25/2023 0 Comments Spore test
One type of test strips for all sterilizers. The spores of both species are found on the test strips used by SMS. Oven sterilization is verified by spores of Bacillus atrophaeus, cultured at 37 C°. Steam and chemical sterilization is verified by the spores of Geobacillus stearothermophis, cultured at 55 C°. Q: Is biological monitoring (spore test strips) acceptable for all methods of sterilization?Ī: Steam, chemical and oven sterilization can all be verified by the dual species test strips used by the SMS at OHSU. In December, you will receive a hard copy of the past years record, along with your certificate of participation in the monitoring service. If you need a copy of your record for a specific period, just call us but your email record of results is satisfactory for verification of compliance. Your records are stored in an encrypted data base on site. 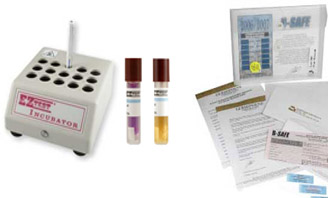
stearothermophis is the test species for steam and chemical sterilizer units. This reduces the chance of a false positive result to essentially zero. They are cultured at 55 C°, too hot for almost every other bacterial species. Geobacillus stearothermophis is a thermophilic bacteria, original isolates in the thermal pools of Yellowstone. Why use two species? Bacillus atrophaeus spores are notoriously resistant to heat, making them perfect for testing heat only sterilizers like ovens. The test strips supplied by the Sterilizer Monitoring Service are imbedded with spores of two species of bacteria Geobacillus stearothermophis and Bacillus atrophaeus. 
Because bacterial spores are the most resistant form of bacteria, spores have been the most common test material. They may not be reprocessed for reuse except by entities that have complied with FDA regulatory requirements and have received FDA clearance to reprocess specific SUDs.Biological monitoring of sterilizer function depends on the sterilizer killing the test organism. Single-use devices (SUDs) are labeled by the manufacturer for a single-use and do not have reprocessing instructions.Cleaning must always be performed prior to sterilization and/or disinfection.Critical items (e.g., surgical instruments) are objects that enter sterile tissue or the vascular system and must be sterile prior to use.Via “Guide to Infection Prevention for Outpatient Settings: Minimum Expectations for Safe Care.” Items Specific To Sterilizer Quality Control Testing sterilization, the only means of confirming sterilization.įor record-keeping compliance, each test must be logged! Testing the parameters of steam penetration leading to sterilization but not confirming sterilizationģ. 
The gages read on the dashboard of the sterilizerĢ. There are three required methods of sterilization monitoring:ġ. However, because spore tests are only done weekly and the results are usually not obtained immediately. What Needs To Be Included In The Logging Of Mechanical Quality Assurance?īiological indicators, or spore tests, are the most accepted means of monitoring sterilization because they assess the sterilization process directly by killing known highly resistant microorganisms. So, what do you need for your weekly spore test when proof of testing is required? It’s an essential piece of equipment in your medical or dental practice. A table-top sterilizer is defined in AAMI ST79’s “Comprehensive Guide To Sterility Assurance” as a compact steam sterilizer that has a chamber volume of less than or equal to 2 cubic feet and that generates its own steam when distilled or deionized water is added by the user.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
